|
4/18/2024 0 Comments Who came up with the atomic theory
Of course we just as easily could have illustrated the law by considering the mass of nitrogen that combines with one gram of oxygen it works both ways!.Note that just as the law of multiple proportions says, the weight of oxygen that combines with unit weight of nitrogen work out to small integers. Line is obtained by dividing the figures the previous line by the smallest O:N ratio in the line above, which is the one for N 2O.But someone who depends solely on experiment would work these out by finding the mass of O that combines with unit mass (1 g) of nitrogen. The numbers in Line are just the mass ratios of O:N, found by dividing the corresponding ratios in line 1.(These numbers were not known in the early days of Chemistry because atomic weights (i.e., molar masses) of most elements were not reliably known.) These ratios were calculated by simply taking the molar mass of each element, and multiplying by the number of atoms of that element per mole of the compound. 
Line shows the ratio of the relative weights of the two elements in each compound.Nitrogen forms a very large number of oxides, five of which are shown here.įigure 2.1.3: Law of Multiple Proportions applied to nitrogen oxides (\(NO_x\)) compounds. It's easy to say this, but please make sure that you understand how it works. In such cases, this law states that the weights of one element that combine with a fixed weight of another of these elements are integer multiples of one another. Many combinations of elements can react to form more than one compound. Scientists did not account for the gases that play a critical role in this reaction. However, Figure 2.1.1 shows that the burning of word does follow the law of conservation of mass. From this observation scientists concluded that mass had been lost. If this law was true, then how could a large piece of wood be reduced to a small pile of ashes? The wood clearly has a greater mass than the ashes. Historically, this was a difficult concept for scientists to grasp. Image as a whole constructed by Jessica Thornton (UCD). Image of ashes courtesy of Walter Siegmund. The law of conservation of mass was formulated by Lavoisier as a result of his combustion experiment, in which he observed that the mass of his original substance-a glass vessel, tin, and air-was equal to the mass of the produced substance-the glass vessel, “tin calx”, and the remaining air.įigure 2.1.1: Image of the wood courtesy of Ehamberg and Stannered on Wikimedia Commons, available under Creative Commons Attribution 2.5 Generic license. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction in other words, mass is conserved. More simply, whatever you do, you will still have the same amount of stuff (however, certain nuclear reactions like fusion and fission can convert a small part of the mass into energy. Antoine Lavoisier (1743-1794) restated this principle for chemistry with the law of conservation of mass, which "means that the atoms of an object cannot be created or destroyed, but can be moved around and be changed into different particles." This law says that when a chemical reaction rearranges atoms into a new product, the mass of the reactants (chemicals before the chemical reaction) is the same as the mass of the products (the new chemicals made). "Nothing comes from nothing" is an important idea in ancient Greek philosophy that argues that what exists now has always existed, since no new matter can come into existence where there was none before. This article explains the theories that Dalton used as a basis for his theory: The theory explains several concepts that are relevant in the observable world: the composition of a pure gold necklace, what makes the pure gold necklace different than a pure silver necklace, and what occurs when pure gold is mixed with pure copper. John Dalton (1766-1844) is the scientist credited for proposing the atomic theory. But how far can this exercise be taken, at least in theory? Can one continue cutting the aluminum foil into halves forever, making smaller and smaller pieces? Or is there some limit, some absolute smallest piece of aluminum foil? Thought experiments like this-and the conclusions based on them-were debated as far back as the fifth century BC. It should be obvious that the pieces are still aluminum foil they are just becoming smaller and smaller. Continue cutting, making smaller and smaller pieces of aluminum foil. Cut one of those smaller pieces in half again. Now there are two smaller pieces of aluminum foil. 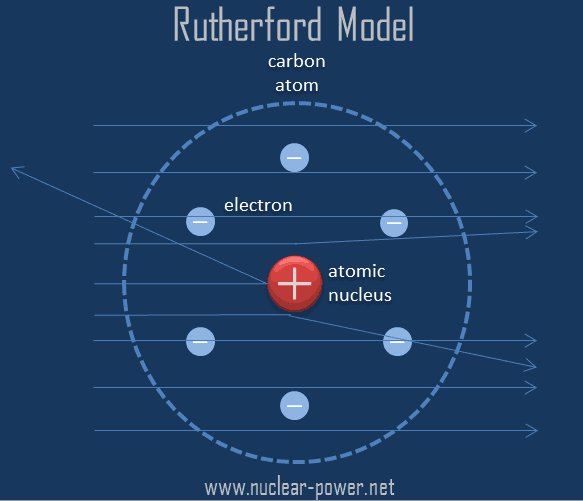
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
